
Please read this in conjunction with the electrolytic compatibility table at the bottom of the page.
When two dissimilar metals or two similar metal that differ only slightly in chemical composition are in contact in the presence of moisture (e.g. a damp or humid atmosphere) electrolytic or galvanic corrosion can occur. This is caused by the generation of a minute currant that causes one of the metals to be eaten away.
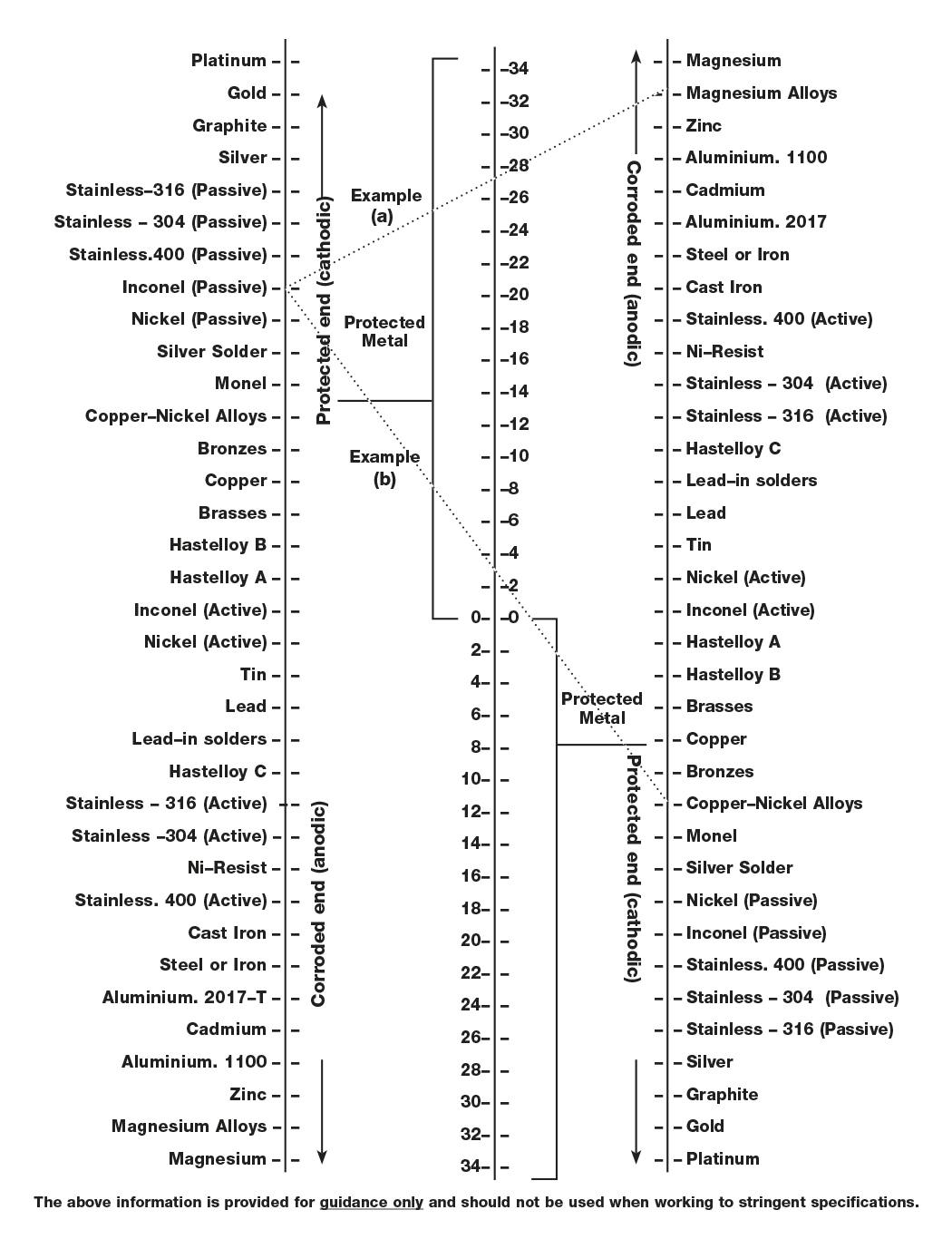
Metals furthest apart in an electrolytic series will corrode most rapidly. The Electrolytic compatibility table shows the possible extent of electrolytic corrosion for comparison purposes, as well as the differences in corrosion rates for metal combinations.
A straight line is drawn between the two metals being considered; one metal selected from the left hand list and the other from the right hand list. The center column number scale represents the degree of possible corrosion activity.
A large number indicated high possibility of electrolytic activity, whilst a low number indicated a better combination for resisting electrolytic corrosion. The left and right hand material columns also indicate which metal will be protected and which will be attacked by corrosion.
Example
A: connecting inconel (passive) on the left scale, with Magnesium Alloys on the right scale, gives a value of 27. This is a high electrolytic risk situation, with the inconel being protected at the expense of the Magnesium Alloy.
B: connecting inconel (passive) with Copper/Nickel alloys gives a value of 3. This is obviously a better corrosion resisting combination.
A greater corrosion rate than indicated can result if a small Anode is in contact with a large Cathode since the electrical current density is increased at the Anode.
Where it is not possible to use materials that will resist corrosion then use a protective agent such as Tef Gel or use Rubber (EPDM) or Nylon washers if the materials are likely to be exposed to particularly corrosive conditions.
Use a ruler to line up the two metals which are being combined. The closer to zero, the lower the risk of galvanic corrosion. The below dashed lines are the examples referred to in the explanation. (a) shows a high risk of galvanic corrosion, where as (b) shows a low risk.
NB: This information is provided as a guide only and Anzor does not accept liability for the application of this information. For advice about stainless steel contact the Nickel Institute.
Fischer DuoPower - One Plug. One Solution. All Materials.
BUMAX® DX 129: The World's Strongest Stainless Steel Bolts
Build without delays with Anzor Quick Drill Screws for timber construction.
To receive useful info and product updates add your details below